Cl charge in coulomb11/23/2023 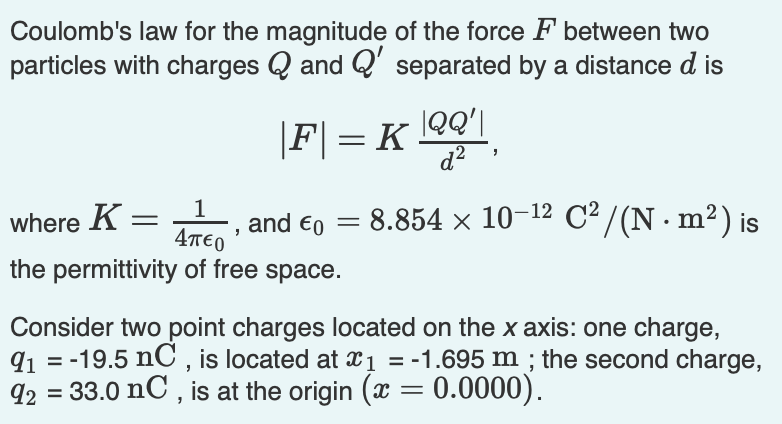 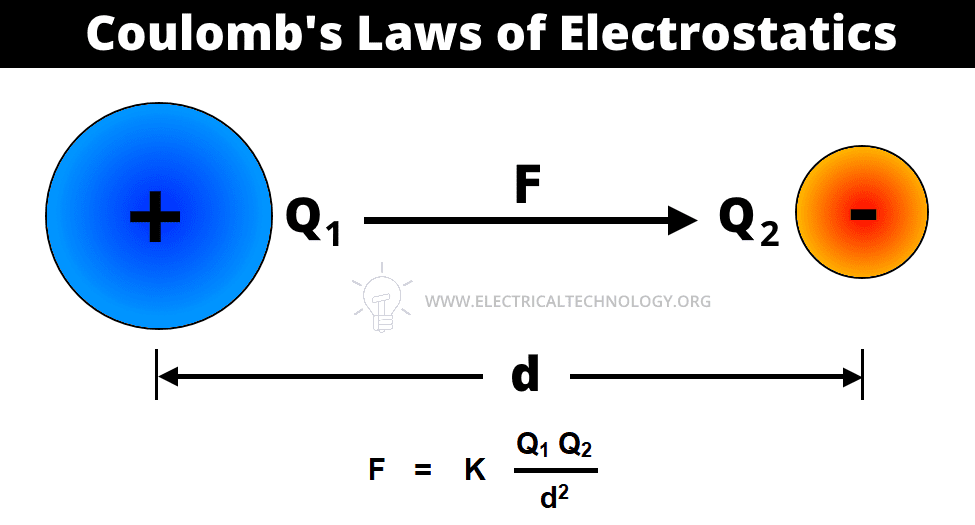
Accumulated charge on you discharges rapidly into the metal, a "sink" for electrons, producing the shock. A common example you've experienced is getting a shock when you touch something metal, usually in the winter when the air is dry. Static electricity refers to charge that doesn't move unless "triggered" to do so. As the flow-chart above shows, we can divide our discussion about charge into static electricity phenomena and electric current. 
In the following sections we'll figure out the details of how charges interact and the units of charge. How many times have you been walking down the street and gotten ejected from Earth by gravity? Gravity has no repulsive component it is a purely attractive force. Forces between charges can be attractive or repulsive. This ought to make you pause for a second because it is already vastly different than the gravitational force, our other invisible force. Those always tend to pair up evenly.Ĭharges exert invisible forces on one another in a specific and predictable way. The universe also tends to balance charges in a given system (a defined piece of the universe), there are generally the same number of positive and negative charges.įor example, in a salt crystal consisting of positively charged sodium ions (Na +) and negatively-charged chloride ions (Cl -), it is highly unlikely that we will have an unpaired charge. When the charge of something changes, it's because it loses or gains a negatively-charged electron. In normal processes (things we would encounter in day-to-day life) charges are neither created from nothing nor destroyed. Objects that are neutral may be that way because of their inherent nature, like the neutron, but more commonly they are neutral because they contain equal numbers of negatively-charged particles (electrons) and positively charged ones (protons).Ĭharge is also a conserved quantity. Matter may be positively-charged, negatively-charged or neutral (not charged). We now know those to be negatively-charged electrons. In more controlled experiments, we can observe that charged objects exert invisible forces on one another.īenjamin Franklin, an early researcher in electricity and charge, assigned the label positive to the charges that tend to move the most. We understand charge because we can observe lightning, see sparking between wires and we can get an electric shock when things like fabrics rub together in the dry air of winter. This means that the most electronegative atom is Fluorine and the least electronegative is Francium.Electric charge, like mass, is a fundamental property of matter. The trend for electronegativity is to increase as you move from left to right and bottom to top across the periodic table. The main properties of an atom dictate it's electronegativity are it's atomic number as well as its atomic radius. The higher the electronegative of an element, the more that atom will attempt to pull electrons towards itself and away from any atom it bonds to. Linus Pauling described electronegativity as “the power of an atom in a molecule to attract electrons to itself.” Basically, the electronegativity of an atom is a relative value of that atom's ability to attract election density toward itself when it bonds to another atom. In 1936, Linus Pauling came up a method for estimating atomic electronegativities forms the basis of our understanding of electronegativity today. To quantify how much ionic character (and how much covalent character) a bond possesses, electronegativity differences between the atoms in the bond can be used. Generally, however, bonds are partially covalent and partially ionic, meaning that there is partial transfer of electrons between atoms and partial sharing of electrons. Only homonuclear bonds are truly covalent, and nearly perfect ionic bonds can form between group I and group VII elements, for example, KF. Most real chemical bonds in nature are neither truly covalent nor truly ionic. Recall that a lowercase Greek delta (\(δ\)) is used to indicate that a bonded atom possesses a partial positive charge, indicated by \(δ^+\), or a partial negative charge, indicated by \(δ^−\), and a bond between two atoms that possess partial charges is a polar bond. \) compares the electron distribution in a polar covalent bond with those in an ideally covalent and an ideally ionic bond.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
